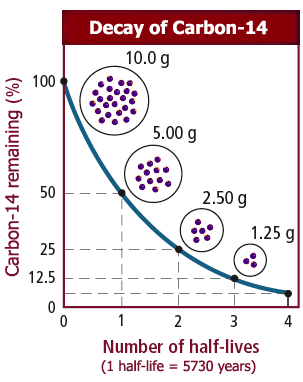
With this formula, we can calculate the amount \(m\) of carbon-14 over the years.Įvery year, the mass \(m\) of carbon-14 is multiplied by \(e^\) decreases by half. Substituting the initial condition \(t = 0\), \(m = 100\) gives \(C = 100\), so Since \(m\) has a continuous decay rate of \(-0.000121\), a general solution to the differential equation is Let's investigate what happens to the sample over time.įirst, we can solve the differential equation. Suppose our sample initially contains 100 nanograms of carbon-14. It turns out that, if the sample is isolated, then \(m\) and \(t\) approximately 4 satisfy the differential equation 
Let \(m\) be the mass of carbon-14 in nanograms after \(t\) years. Suppose we have a sample of a substance containing some carbon-14. 
It is naturally produced in the atmosphere by cosmic rays (and also artificially by nuclear weapons), and continually decays via nuclear processes into stable nitrogen atoms. Content Radioactive decay and half-life Decay of carbon-14Ĭarbon-14 is a radioactive isotope of carbon, containing 6 protons and 8 neutrons, that is present in the earth's atmosphere in extremely low concentrations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
